injection the effects were much stronger. In earlier animal model studies of mRNA vaccines, such administration was clearly preferred over a straight i.v. The big surface vein in that region is the cephalic vein, and it's down along where the deltoid and pectoral muscles meet, not high up in the shoulder. That's why a muscle like the deltoid is preferred, because it's a good target of thicker muscle tissue without any easily hit veins or arteries at the site of injection. The injection is intramuscular, not into the bloodstream. Compare this, though, to what happens in vaccination. 
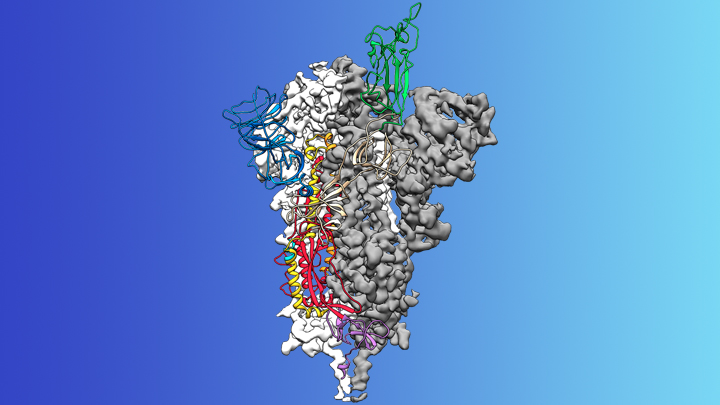
They then have a clear shot at the endothelial cells lining the airway vasculature, which are the very focus of these two new papers. The viral infection process leads at the end to lysis of the the host cell and subsequent dumping of a load of new viral particles - and these get dumped into the cellular neighborhood and into the bloodstream. We know now that the huge majority of such infections are spread by inhalation of virus-laden droplets from other infected people, so the route of administration is via the nose and/or lungs, and the cells lining your airway are thus the first ones to get infected. Consider what happens when you're infected by the actual coronavirus. There are several reasons why the situation is different. So I've been getting questions about what this means for vaccination: if we're causing people to express Spike protein via mRNA or adenovirus vectors, are we damaging them just as if they'd been infected with coronavirus? Fortunately, the answer definitely seems to be "no" - in fact, the pseudovirus paper notes near the end that the antibody response generated by vaccination against the Spike protein will be beneficial in two ways, against infection and against the Spike-mediated endothelial damage as well. That happens as the coronvirus infects vascular tissue, of course, but this work shows that it's not the whole process of viral infection that's responsible for all the trouble: it starts with the initial binding event. The response to the pseudovirus was quite different in these two, suggesting that it is indeed the binding of the Spike protein to ACE2 that's a key part of this process. The pseudovirus team went on to compare endothelial cells with different mutational forms of the ACE2 surface protein (S680D, with increased stability and S680L, with decreased stability). In the first study, hamsters were injected with a pseudovirus was created that expressed surface Spike protein, while in the second the researchers just injected the protein directly into mice. 
These are almost certainly looking at the same phenomena - the lungs are of course full of vascular tissue, and what's being seen in both cases is very likely mediated by effects on the vascular endothelium. 
There's been a recent report about the vascular effects of the Spike protein alone (not coronavirus infection per se), and another presentation on similar effects in lung tissue. I've been getting a lot of questions in the last few days about several Spike-protein-related (and vaccine-related) topics, so I thought this would be a good time to go into them. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
